
Earning her doctoral degree in an era when science seems to have been engulfed by the culture wars has made LaBella think it’s time for scientists to be more “visible” and to better translate their findings for the public. Her road trip buddy, Abigail LaBella, just defended her PhD in genetic and genomic research and carried a sign with a green cow and lab bench that read “The Scientific Method: You have NOT died of smallpox”). Thank a scientist!“) might contribute to at least one person realizing that not all scientists are “part of a huge conspiracy.” She hoped her presence (and maybe her sign “Got Plague? Yeah, me neither. “As a minority woman in the sciences, right now is a tough time in terms of identity attacks, and it’s important to bolster my sense of confidence and this seems to be a good way to do that.” “I just love the idea of being surrounded by so many scientists and like-minded people,” said Wilson, who is researching hydrology and water resources. 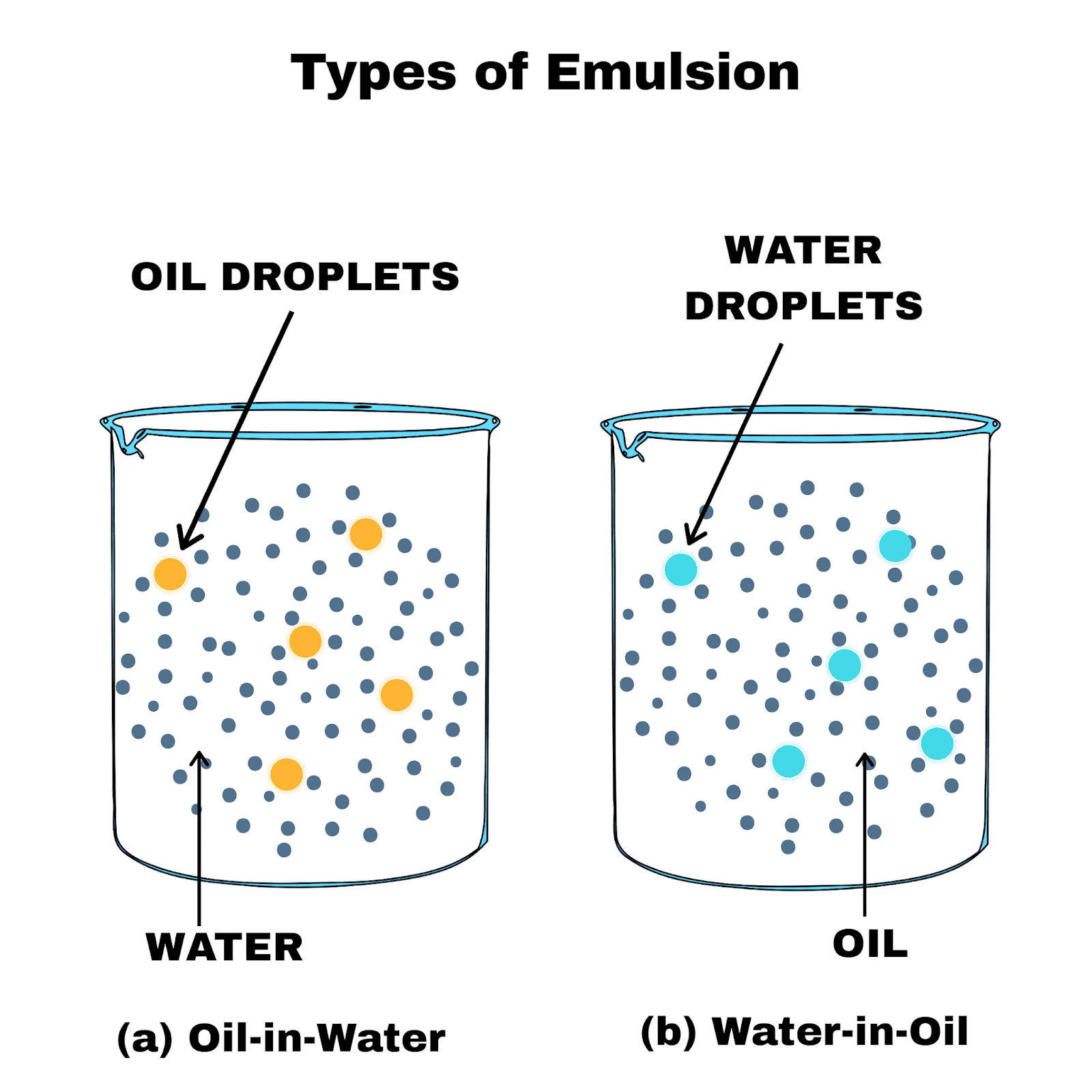
The encouraging vibe drew PhD student Tiffany Wilson from Duke University. One emblematic sign read: “Think like a proton. “Realistically, I can’t say I have much hope but I hope the rest of us who are not here will see how important it is and will get on board.”Īmong the crowd mobbing the central stage, the sense of hope belied the weather. She acknowledged the march might not make a difference nationally but was still glad they braved the weather. Her kids, Brayden and Addison, toted homemade signs (which included an angry beaker and an inspiring Barack Obama quote: “We are the change that we seek.”). It’s not just a nerdy thing anymore,” said Danielle Heath. “I think that it is important they see the love for science, the support for science. The lead protestors carried a 3-½ lane-wide “March for Science” banner.ĭanielle Heath and husband Chris brought 2 of her 3 kids from their home near Washington DC. For 4 hours they rooted, railed and rallied, and then they marched down Constitution Avenue to Capitol Hill. They carried signs: “There is no Planet B,” “Remember Polio? Me Neither,” “I can march thanks to science > new hip.” And even as the rain pelted them, they cheered a rock concert stage that included Bill Nye the Science Guy, musician Thomas Dolby (of the 1982 hit song “She Blinded Me with Science”), poet Jane Hershfield and others. They sported crocheted pink “brain hats.” They brought their kids. They came from Buffalo, small towns in Maine and Maryland, from Durham, North Carolina and points west. Watch this video to learn more about chemistry.WASHINGTON DC-Sometimes drizzling and sometimes pummeling, H2O couldn’t dampen the spirits of thousands of scientists and supporters who flocked to Washington DC for Saturday’s March for Science-one of 600 such events taking place worldwide.įor hours, they streamed through security checkpoints at the base of the Washington Monument. The non-polar tail dissolves in the oil droplets by stabilizing the emulsion to form a micelle above the critical micelle concentration of the soap solution. When soap solution is added to an oil-in-water emulsion, the polar head dissolves in the water phase. The polar head is hydrophilic (water-loving) in nature, and the non-polar tail is hydrophobic (oil-loving). For example, soap molecules have a polar head and a non-polar hydrocarbon tail. Soaps and detergents are the most commonly used emulsifiers. The emulsions, therefore, need to be stabilized.Īn emulsifying agent, also called an emulsifier, stabilizes the emulsion by lowering the interfacial tension between the two immiscible liquids. But these emulsions are thermodynamically unstable because the dispersed droplets simultaneously come together and form separate layers. The second “water in oil” emulsion, in which the water is the dispersed phase and oil is the dispersion medium.Įmulsions can be obtained by vigorously agitating a mixture of both liquids. For example, oil and water can form the first “oil in water” emulsion, in which the oil is the dispersed phase, and water is the dispersion medium. Two liquids can form different types of emulsion. An emulsion is a colloidal system in which the dispersion phase and the dispersed medium (also known as the continuous phase) are in the form of liquids normally immiscible in nature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
